Please choose your shipping country in order to get correct prices and freight cost.
Africa
America
Asia
Europe
Oceania
-
Africa
- Algeria
- Angola
- Benin
- Botswana
- Burkina Faso
- Burundi
- Cameroon
- Cap Verde
- Central African Republic
- Chad
- Comoros
- Congo, Dem. Rep.
- Congo, Repub. of the
- Cote d'Ivoire
- Djibouti
- Egypt
- Equatorial Guinea
- Eritrea
- Ethiopia
- Gabon
- Gambia
- Ghana
- Guinea
- Guinea Bissau
- Kenya
- Lesotho
- Liberia
- Libya
- Madagascar
- Malawi
- Mali
- Mauritania
- Mauritius
- Mayotte
- Morocco
- Mozambique
- Namibia
- Niger
- Nigeria
- Reunion
- Rwanda
- Saint Helena
- Sao Tome & Principe
- Senegal
- Seychelles
- Sierra Leone
- Somalia
- South Africa
- Sudan
- Swaziland
- Tanzania
- Togo
- Tunisia
- Uganda
- Western Sahara
- Yemen
- Zambia
- Zimbabwe
-
America
- Anguilla
- Antigua & Barbuda
- Argentina
- Aruba
- Bahamas
- Barbados
- Belize
- Bermuda
- Bolivia
- Brazil
- British Virgin Is.
- Canada
- Cayman Islands
- Chile
- Colombia
- Costa Rica
- Cuba
- Dominica
- Dominican Republic
- Ecuador
- El Salvador
- French Guiana
- Grenada
- Guadeloupe
- Guatemala
- Guyana
- Haiti
- Honduras
- Jamaica
- Martinique
- Mexico
- Montserrat
- Netherlands Antilles
- Nicaragua
- Panama
- Paraguay
- Peru
- Puerto Rico
- Saint Kitts & Nevis
- Saint Lucia
- Saint Vincent & the Grenadines
- St Pierre & Miquelon
- Suriname
- Trinidad & Tobago
- Turks & Caicos Is
- United States
- Uruguay
- Venezuela
- Virgin Islands
-
Asia
- Afghanistan
- Armenia
- Azerbaijan
- Bahrain
- Bangladesh
- Bhutan
- Brunei
- Burma
- Cambodia
- China
- East Timor
- Gaza Strip
- Georgia
- Hong Kong
- India
- Indonesia
- Iran
- Iraq
- Israel
- Japan
- Kazakhstan
- Korea, North
- Korea, South
- Kuwait
- Kyrgyzstan
- Laos
- Lebanon
- Macau
- Malaysia
- Maldives
- Mongolia
- Nepal
- Oman
- Pakistan
- Philippines
- Qatar
- Saudi Arabia
- Singapore
- Sri Lanka
- Syria
- Taiwan
- Tajikistan
- Thailand
- Turkmenistan
- United Arab Emirates
- Uzbekistan
- Vietnam
- Yemen
-
Europe
- Albania
- Andorra
- Austria
- Belarus
- Belgium
- Bosnia & Herzegovina
- Bulgaria
- Croatia
- Cyprus
- Czech Republic
- Denmark
- Estonia
- Faroe Islands
- Finland
- France
- Germany
- Gibraltar
- Greece
- Greenland
- Guernsey
- Hungary
- Iceland
- Ireland
- Isle of Man
- Italy
- Jersey
- Latvia
- Liechtenstein
- Lithuania
- Luxembourg
- Macedonia
- Malta
- Moldova
- Monaco
- Netherlands
- Norway
- Poland
- Portugal
- Romania
- Russia
- San Marino
- Serbia
- Slovakia
- Slovenia
- Spain
- Sweden
- Switzerland
- Turkey
- Ukraine
- United Kingdom
- Oceania
- Home
- Products
-
Products
- Pharmaceutical Quality Dextran
- Dextran 1 (EP/USP)
- Dextran 1.5
- Dextran 3.5
- Dextran 5
- Dextran 10
- Dextran 20
- Dextran 25
- Dextran 40 (EP/USP/JP)
- Dextran 60 (EP)
- Dextran 70 (EP/USP/JP)
- Dextran 110
- Dextran 150
- Dextran 250
- Dextran 500
- Technical Quality Dextran
- Dextran T1
- Dextran T1.5
- Dextran T3.5
- Dextran T5
- Dextran T6
- Dextran T10
- Dextran T20
- Dextran T25
- Dextran T40
- Dextran T60
- Dextran T70
- Dextran T110
- Dextran T150
- Dextran T250
- Dextran T500
- Dextran T750
- Dextran Derivatives
- DEAE-Dextran 500
- DEAE-Dextran 500 Pharmaceutical Quality
- Dextran Ultra
- Dextran 10
- Dextran 20
- Dextran 40
- Dextran 70
- Dextran 500
- Dextran Standards & GPC Standards
- Dextran Kit
- Dextran 1
- Dextran 5
- Dextran 12
- Dextran 25
- Dextran 50
- Dextran 80
- Dextran 150
- Dextran 270
- Dextran 410
- Dextran 670
- Dextran 2000
- Dextran 3800
- Dextran 5400
- Application Areas
- about dextran
- PentaHibe®
DEAE-Dextran
Pharmacosmos DEAE-Dextran are based on Pharmacosmos’ world-leading Dextran products, and Pharmacosmos DEAE-Dextran are also produced at Pharmacosmos’ state-of-the-art, GMP-approved facility in Denmark.
As a natural consequence of Pharmacosmos’ commitment to quality, Pharmacosmos DEAE-Dextran are developed and manufactured according to the same advanced standards and requirements of the highest quality as applied in the development and manufacturing of our world-leading Dextran products.
DEAE-Dextran
DEAE-Dextran is made by reaction of dextran with (2-chloroethyl) diethylammoniumchlorid in alkaline solution.
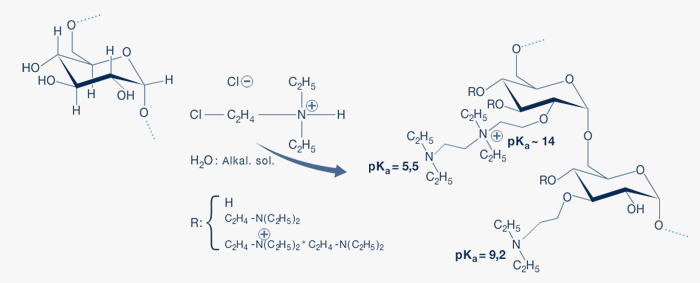
Fig. 1: DEAE-Dextran
DEAE-Dextran is a polycation. DEAE - Dextran contains three basic groups with different pKa values. There are two tertiary groups, one in the single substituted glucose unit having a pKa value of approximately 9.2 and the tertiary group in the tandem substitution having a pKa value of approximately 5.5. The quaternary amino group in the tandem substitution has a pKa value ~ 14.
Below in Fig.2 is shown the titration curve from titration of DEAE - Dextran after alkanation.
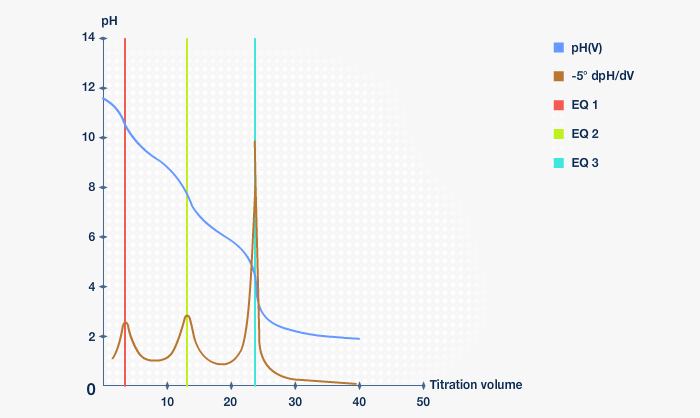
Fig. 2: Titration after alkanation
The red vertical bar shows at which point the added sodium hydroxide is titrated. The green vertical bar shows when the tertiary group with pKa of approximately 9.2 is titrated, and finally the blue vertical bar shows when the tertiary group with pKa near 5.5 is titrated.
The total amount of nitrogen can be calculated from the titration curve or by Kjeldahl-analysis.
The nitrogen content of approximately 3.2 % is equivalent to one charged group per three glucose units.
As seen in Fig.1 DEAE-Dextran contains more tertiary groups than quaternary amino groups. Calculated on the amount of nitrogen in the DEAE-Dextran, approximately 2/3 of the nitrogen content belongs to the tandem substitution.
DEAE-Dextran is stable in solution at ambient temperatures and pH values ranging from pH = 4 to pH =14. The dry powder has good stability.

Contact Specialist
Anne-Marie J. Ørkild
Director, International Sales and Business Carbohydrates
Phone: +45 5948 5959
E-mail: dextran@pharmacosmos.com
Approved
Pharmacosmos is the only dextran manufacturer holding certificates from the US FDA and European Directorate for the Quality of Medicines (EDQM)
Quality Delivered
Pharmacosmos delivers a unique dextran quality through a production technology that completely avoids the use of organic solvents and a quality system that meets the strictest cGMP standards for human medicines
Global Service

Pharmacosmos sells and ships directly to clients everywhere in the World. We deliver Pharmaceutical Quality Dextran of the highest standards, including those of the European Pharmacopoeia (EP), the United States Pharmacopoeia (USP), and the Japanese Pharmacopoeia (JP).

